Luxturna: costly but welcomed
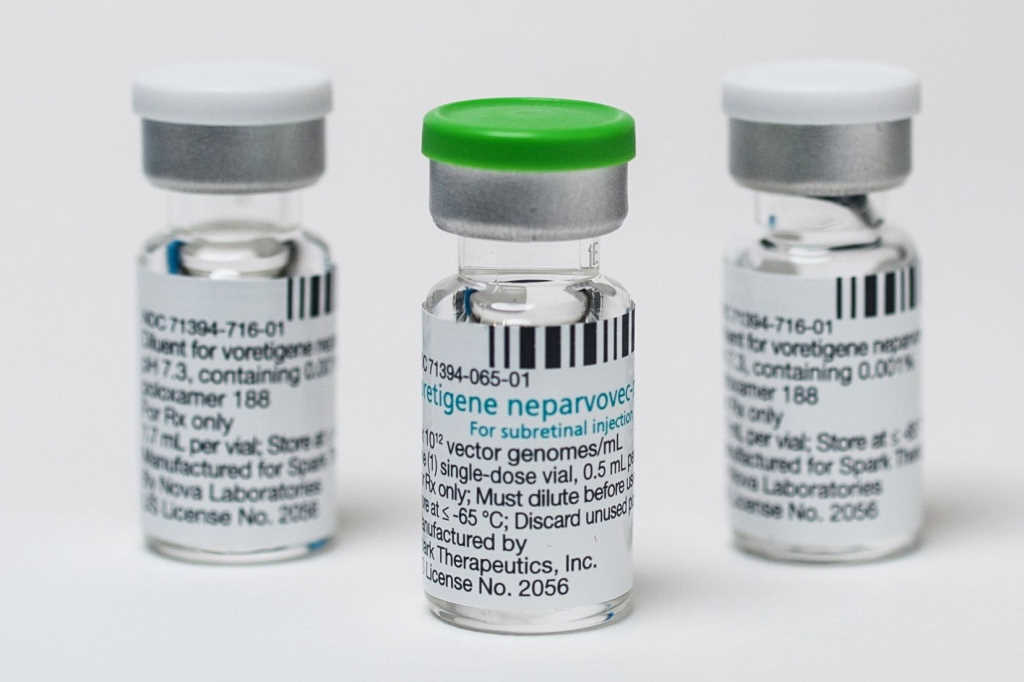
The US Food and Drug Administration (FDA) has fast-tracked and approved a novel gene therapy, Luxturna (voretigene neparvovec-rzyl), to treat patients with a rare form of inherited vision loss that can result in blindness.
The news was welcomed by advocacy group Retina International and international retina specialists, including Auckland-based Associate Professor Andrea Vincent.
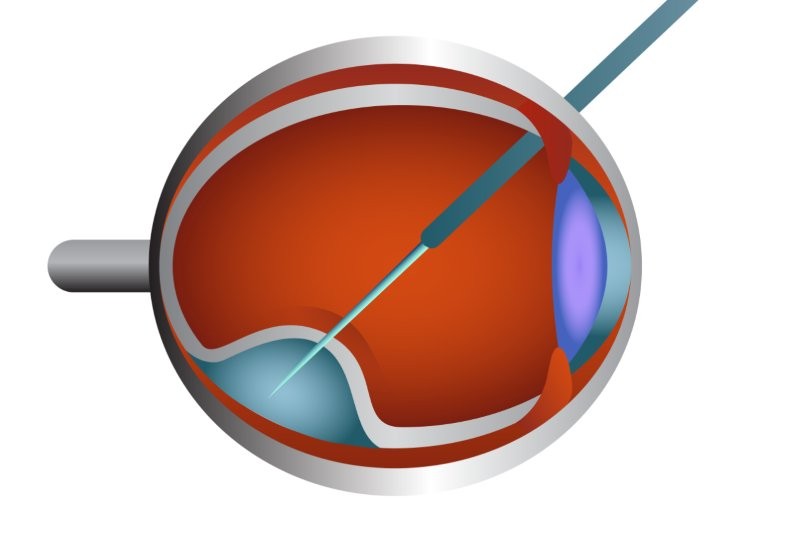
“This represents a very exciting milestone in the treatment of inherited retinal diseases,” said A/Prof Vincent. “There is a complex surgical procedure that has been developed to deliver this gene therapy. Therefore, it is likely at this point, that any patient would have to travel overseas to receive this treatment, although expertise exists in Australia.”
Luxturna has been approved in the US for the treatment of patients with confirmed biallelic RPE65 mutation-associated retinal dystrophy that leads to vision loss and may cause complete blindness. The condition is rare, with approximately 1,000 to 2,000 patients believed to suffer from it in the US and just a few in New Zealand. Only two individuals have been identified in the New Zealand Database of Inherited Retinal Diseases, confirmed A/Prof Vincent.
Though the new treatment was welcomed, many international commentators however, are questioning the cost of treatment. Manufacturer Spark Therapeutics announced the cost at a whopping US$425,000 per eye at the end of last year, not including surgery costs, reigniting the cost of medicines debate in the US.
“(In New Zealand) application would have to be made on a case-by-case basis,” said A/Prof Vincent. “However, as RPE65 retinal disease affects very young children, the socioeconomic cost of supporting and educating severely visually impaired children for the duration of their life is not insignificant, and therefore this likely represents a cost benefit.”
But the cost shouldn’t detract from what is an incredible scientific breakthrough, she added. “This is a very exciting advancement for the treatment of inherited retinal diseases and the lessons learnt from [the trials] have allowed many other gene therapies for retinal degenerations to advance more rapidly.”
The RPE65 gene provides instructions for making an enzyme that is essential for normal vision. Thus, Luxturna works by delivering a normal copy of the RPE65 gene directly to retinal cells using a naturally occurring adeno-associated virus, which has been modified using recombinant DNA techniques. These retinal cells then produce the protein that converts light to an electrical signal in the retina to restore patient’s vision loss.
“[This] approval marks another first in the field of gene therapy — both in how the therapy works and in expanding the use of gene therapy beyond the treatment of cancer to the treatment of vision loss,” said FDA commissioner Dr Scott Gottlieb in a statement. “I believe gene therapy will become a mainstay in treating, and maybe curing, many of our most devastating and intractable illnesses.”
Congratulating those involved in Luxturna’s development, Christina Fasser, president of Retina International, said the news marks the beginning of “an exciting and hopeful new era in the development of treatments.”
More about this incredible new treatment will be revealed at the Retina International meeting in Auckland this month (see box) as one of the keynote speakers is Dr Jean Bennett, a lead researcher on Luxturna and an ophthalmologist at the University of Pennsylvania’s Perelman School of Medicine. Others senior researchers involved in Luxturna’s development include Dr Bennett’s husband, Dr Albert Maguire, an ophthalmologist at The Children's Hospital of Philadelphia (CHOP) and Dr Katherine High, president and chief scientific officer at Spark Therapeutics.
Retina 2018 in Auckland
The 20th Retina International World Congress is being held in the Owen G Glenn Building at the University of Auckland from 10-11 February. The Congress will bring together some of the world’s foremost retinal scientists and clinicians along with global leaders in patient advocacy and peer support. It is aimed at anyone interested retinal disorders.
The scientific programme, organised by Associate Professor Andrea Vincent, covers two days with a variety of sessions, including keynote addresses given by leading international researchers, plenary sessions, patient-focused sessions and education for healthcare professionals and scientists.
For more and to register, go to http://retina2018newzealand.com