Longer-lasting anti-VEGF therapies
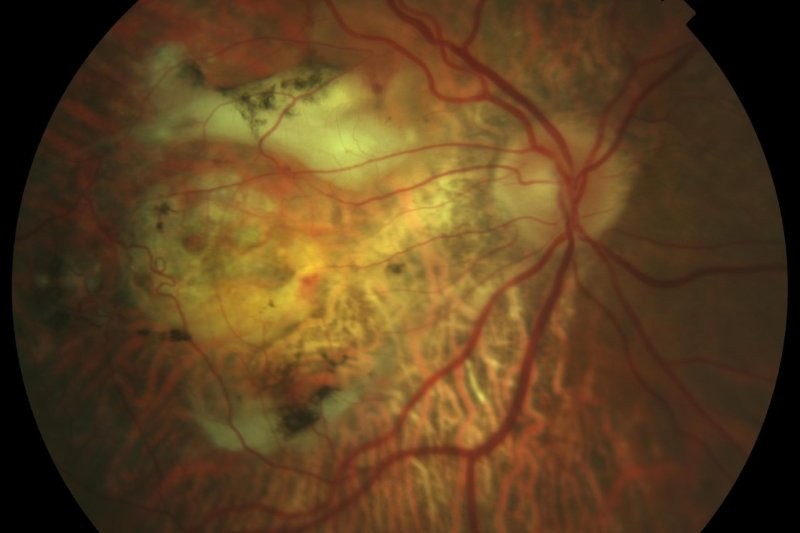
Anti-vascular endothelial growth factor (anti-VEGF) therapy has transformed the management of retinal diseases, such as neovascular age-related macular degeneration (nAMD), diabetic macular oedema (DMO) and retinal vein occlusion (RVO) over the past two decades. By inhibiting pathological neovascularisation and reducing vascular permeability, anti-VEGF injections have preserved vision for millions of patients worldwide. However, the need for frequent intravitreal injections remains a significant burden for patients and healthcare systems.
History
The discovery of bevacizumab (Avastin) as a retinal therapy was serendipitous. Initially developed and approved in 2004 as an intravenous treatment for metastatic colorectal cancer, ophthalmologist Dr Philip Rosenfeld at the Bascom Palmer Eye Institute, US, observed that patients with nAMD and cancer had an improvement in their macular disease when treated with intravenous Avastin. In 2005–2006, pioneering clinicians administered intravitreal Avastin off-label, rapidly demonstrating significant reductions in macular oedema and improvements in visual acuity. Subsequently, ranibizumab (Lucentis) – a monoclonal antibody fragment derived from bevacizumab and specifically designed for ophthalmic use – was approved in 2006 following two highly successful randomised clinical trials (Marina and Anchor). Due to cost, many countries, including New Zealand, continued to use Avastin off-label for ophthalmic indications. This decision was justified by the Catt trial, which showed no significant difference in efficacy between Avastin and Lucentis.
Aflibercept (Eylea) was the next significant development. Both Lucentis and Avastin bind and inhibit VEGF-A, but Eylea is a fusion protein that binds VEGF-A, VEGF-B and placental growth factor. The hope is that with broader VEGF blockage, Eylea would be able to better control macular exudation, leading to better-sustained action. This is supported by the results of the View-1 and View-2 nAMD studies showing that visual acuity is preserved with Eylea injections every eight weeks compared with Lucentis every four weeks, with similar anatomical improvement. However, under careful observation, there is a ‘saw-tooth’ pattern to the central macular thickness plot for Eylea every eight weeks in the View-1 and 2 studies, with a trend of increasing thickness at the eight-week mark after an Eylea injection1. This suggests not all patients on Eylea every eight weeks have complete suppression of the exudation process, a finding mirrored in our clinical experience.
Currently, biosimilars to both Lucentis and Eylea are available and several are FDA-approved. Many countries employ biosimilars to give patients access to a similar visual outcome to Lucentis and Eylea at a fraction of the price. Biosimilars are not yet widely available in New Zealand.
Treatment delivery innovation
Further to drug innovation, there is also innovation in treatment delivery. It was recognised that not all patients require ongoing regular injections every four weeks, as suggested in Marina and Anchor1. Many patients may be able to have fewer injections while maintaining adequate control. Pro re nata (PRN) treatment, in which injections were given only when fluid was present, was attempted, but patients would still need to be seen every four weeks and any recurrence of exudation could lead to further macular damage and thus worsen visual outcomes. Instead, patients are now treated proactively with a treat-and-extend regimen, in which an injection is given at every visit, with the interval lengthened if there is no exudation and shortened if there is. This approach reduces the number of injections required, compared with fixed dosing, while maintaining acuity gain2,3.
Although a treat-and-extend protocol reduces treatment burden, patients still need frequent follow-up. Multiple studies have highlighted that a substantial proportion of patients with retinal diseases experience delayed or missed appointments in routine practice, often due to comorbidities, logistical challenges and the cumulative strain of frequent injections. These issues affect around 10–30% of patients and are associated with worse anatomical and visual outcomes4,5.
Newer drugs
New molecules have emerged to address the unmet need for sustained retinal drying and stable visual outcomes over fewer injections. Brolucizumab (Beovu), FDA-approved in 2019, is a small single-chain antibody fragment against VEGF-A, designed to extend durability due to its high molar concentration. Early data showed strong anatomical drying and the potential for 12-week intervals. However, post-marketing reports of occlusive retinal vasculitis and vascular inflammation significantly limited its adoption and it is nowadays rarely used due to safety concerns6.
Building on the success of Eylea, Aflibercept 8mg (Eylea HD) was developed and approved by the FDA in 2023 for ophthalmic use, with the Pulsar and Proton studies supporting its efficacy and improved durability7,8.
Faricimab (Vabysmo), approved in 2022, introduced dual inhibition of VEGF-A and angiopoietin-2. This bispecific antibody has demonstrated improved durability and fluid control compared with earlier agents, with many patients achieving stable disease on 12- to 16-week intervals. Real-world experience continues to support its efficacy and safety9.
Moreover, the pivotal trials for each agent used different re-treatment criteria, limiting the validity of naïve cross-trial comparisons. When these methodological differences are taken into account through indirect comparative analyses, the data suggest Vabysmo may provide a longer duration of action than Eylea HD10. In New Zealand, Vabysmo was Pharmac-funded for patient use in December 2025, offering a meaningful step towards extended durability, albeit under very restrictive special authority criteria.

Non-responders
Non-responders or suboptimal responders are patients who present with persistent intraretinal or subretinal fluid despite regular treatment, minimal visual acuity gain, or an inability to extend beyond short treatment intervals without recurrence of exudation. From a therapeutic perspective, switching between agents can improve anatomical drying in some cases, likely due to broader pathway inhibition (for example, VEGF-B and placental growth factor blockade with aflibercept, or dual VEGF-A and angiopoietin-2 inhibition with faricimab11). However, even with newer molecules designed for greater durability, a subset of patients continues to require four-weekly treatment to maintain disease control.
Future treatments
Several new therapies are on the horizon, driven by the need to reduce treatment burden and enhance long-term outcomes. Tyrosine kinase inhibitors, gene therapy, novel sustained-delivery devices and next-generation biologics may further extend dosing intervals. As research continues to advance, it is expected that additional emerging therapies will further enhance treatment longevity and management options for patients with retinal diseases. Tyrosine kinase-based anti-angiogenic approaches are formulated in biodegradable nanoparticles designed for six to 12-monthly intravitreal dosing. Gene therapy for neovascular disease is designed to deliver a gene vector encoding anti-VEGF to retinal cells, thereby reducing the need for continuous injections, even though they would still need to be repeated. Sustained-delivery devices physically release a therapeutic agent over many months. The Port Delivery System showed a mean time to first refill of ~15 months, with visual outcomes similar to those with monthly injections12-15.
The situation in New Zealand
Access to newer anti-VEGF is limited for New Zealanders with macular diseases. Vabysmo is currently only Pharmac-funded for patients with nAMD or DMO who failed Avastin treatment and who have not been treated with Eylea for more than three months. However, there are many patients with these diseases who do not adequately respond to Eylea or require Eylea every four to six weeks. Because of the current funding setting, they are denied access to a more effective drug. Furthermore, patients with RVO are currently not Pharmac-funded for either Eylea or Vabysmo and thus only have access to off-label Avastin for their ophthalmic disease. Future Pharmac applications to broaden access to Vabysmo for patients with nAMD and RVO are planned.
The optometrist’s role
The optometrist plays an important role in both identifying new patients with macular disease requiring treatment and detecting reactivation in patients on treatment when the interval is extended or they are lost to follow-up. If a macular disease requiring treatment is detected, it is important to make an urgent referral and see the patient as soon as possible, to give the best chance of salvaging vision. Furthermore, injection fatigue is a real problem for long-term patients. It is important for optometrists to explain the need for long-term therapy and to encourage patients to continue their treatment. While injections can be distressing, they remain essential for preserving vision.
References
- Hang A, Feldman S, Amin AP, Ochoa JAR, Park SS. Intravitreal Anti-Vascular Endothelial Growth Factor Therapies for Retinal Disorders. Pharmaceuticals (Basel). 2023 Aug 11;16(8):1140. doi: 10.3390/ph16081140. PMID: 37631054; PMCID: PMC10458692.
- Oubraham H, Cohen SY, Samimi S, Marotte D, Bouzaher I, Bonicel P, Fajnkuchen F, Tadayoni R. Inject and extend dosing versus dosing as needed: a comparative retrospective study of ranibizumab in exudative age-related macular degeneration. Retina. 2011 Jan;31(1):26-30.
- Brown DM, Regillo CD. Anti-VEGF agents in the treatment of neovascular age-related macular degeneration: applying clinical trial results to the treatment of everyday patients. Am J Ophthalmol. 2007 Oct;144(4):627-37.
- Gillies MC, Hunyor AP, Arnold JJ, Guymer RH, Wolf S, Pecheur FL, Munk MR, McAllister IL. Macular Atrophy in Neovascular Age-Related Macular Degeneration: A Randomized Clinical Trial Comparing Ranibizumab and Aflibercept (RIVAL Study). Ophthalmology. 2020 Feb;127(2):198-210.
- Khurana RN, Wang JC, Zhang S, Li C, Lum F. Loss to Follow up in Patients with Proliferative Diabetic Retinopathy Treated with Anti-VEGF Therapy and/or Panretinal Photocoagulation in the United States. Ophthalmol Retina. 2024;8(10):953-961.
- Bilgic A, Kodjikian L, March de Ribot F, Vasavada V, Gonzalez-Cortes JH, Abukashabah A, Sudhalkar A, Mathis T. Real-World Experience with Brolucizumab in Wet Age-Related Macular Degeneration: The REBA Study. J Clin Med. 2021 Jun 23;10(13):2758.
- Brown DM, Boyer DS, Do DV, Wykoff CC, Sakamoto T, Win P, Joshi S, Salehi-Had H, Seres A, Berliner AJ, Leal S, Vitti R, Chu KW, Reed K, Rao R, Cheng Y, Sun W, Voronca D, Bhore R, Schmidt-Ott U, Schmelter T, Schulze A, Zhang X, Hirshberg B, Yancopoulos GD, Sivaprasad S; PHOTON Investigators. Intravitreal aflibercept 8 mg in diabetic macular oedema (PHOTON): 48-week results from a randomised, double-masked, non-inferiority, phase 2/3 trial. Lancet. 2024 Mar 23;403(10432):1153-1163.
- Lanzetta P, Korobelnik JF, Heier JS, Leal S, Holz FG, Clark WL, Eichenbaum D, Iida T, Xiaodong S, Berliner AJ, Schulze A, Schmelter T, Schmidt-Ott U, Zhang X, Vitti R, Chu KW, Reed K, Rao R, Bhore R, Cheng Y, Sun W, Hirshberg B, Yancopoulos GD, Wong TY; PULSAR Investigators. Intravitreal aflibercept 8 mg in neovascular age-related macular degeneration (PULSAR): 48-week results from a randomised, double-masked, non-inferiority, phase 3 trial. Lancet. 2024 Mar 23;403(10432):1141-1152.
- Heier JS, Khanani AM, Quezada Ruiz C, Basu K, Ferrone PJ, Brittain C, Figueroa MS, Lin H, Holz FG, Patel V, Lai TYY, Silverman D, Regillo C, Swaminathan B, Viola F, Cheung CMG, Wong TY; TENAYA and LUCERNE Investigators. Efficacy, durability, and safety of intravitreal faricimab up to every 16 weeks for neovascular age-related macular degeneration (TENAYA and LUCERNE): two randomised, double-masked, phase 3, non-inferiority trials. Lancet. 2022 Feb 19;399(10326):729-740.
- Grimaldi G, Cancian G, Rizzato A, Casanova A, Perruchoud-Ader K, Clerici M, Consigli A, Menghini M. Intravitreal faricimab for neovascular age-related macular degeneration previously treated with traditional anti-VEGF compounds: a real-world prospective study. Graefes Arch Clin Exp Ophthalmol. 2024 Apr;262(4):1151-1159.
- Rothbächer J, Khalil H, Eidherr M, Bolz M. Additional Effects of Faricimab in Aflibercept Low-Responders: Retinal Morphology and Function in Eyes with Neovascular Age Related Macular Degeneration Following a Switch Between Two Anti-VEGF Agents. Clin Ophthalmol. 2025 Sep 3;19:3145-3152. doi: 10.2147/OPTH.S530355. PMID: 40923045; PMCID: PMC12414467.
- Gonzalez-Cortes JH, Martinez-Pacheco VA, Gonzalez-Cantu JE, Bilgic A, March de Ribot F, Sudhalkar A, Mohamed-Hamsho J, Kodjikian L, Mathis T. Current Treatments and Innovations in Diabetic Retinopathy and Diabetic Macular Edema. Pharmaceutics. 2023; 15(1):122.
- Chandra S, Tan EY, Empeslidis T, Sivaprasad S. Tyrosine Kinase Inhibitors and their role in treating neovascular age-related macular degeneration and diabetic macular oedema. Eye (Lond). 2023 Dec;37(18):3725-3733.
- Campochiaro PA, Avery R, Brown DM, Heier JS, Ho AC, Huddleston SM, Jaffe GJ, Khanani AM, Pakola S, Pieramici DJ, Wykoff CC, Van Everen S. Gene therapy for neovascular age-related macular degeneration by subretinal delivery of RGX-314: a phase 1/2a dose-escalation study. Lancet. 2024 Apr 20;403(10436):1563-1573.
- Holekamp NM, Campochiaro PA, Chang MA, Miller D, Pieramici D, Adamis AP, Brittain C, Evans E, Kaufman D, Maass KF, Patel S, Ranade S, Singh N, Barteselli G, Regillo C; all Archway Investigators. Archway Randomized Phase 3 Trial of the Port Delivery System with Ranibizumab for Neovascular Age-Related Macular Degeneration. Ophthalmology. 2022 Mar;129(3):295-307.

Dr Francesc March de Ribot is an ophthalmologist specialising in surgical retina, glaucoma and complex cataract and a researcher at the University of Otago.

Dr Leo Sheck specialises in medical retina, genetic eye disease, electrodiagnostics for complex retina and optic nerve diseases and cataract surgery, especially with co-existing retinal diseases. He consults for Te Whatu Ora Auckland and Retina Specialists and is a director of the NZ Save Sight Society.