TearCare for MGD bags FDA approval
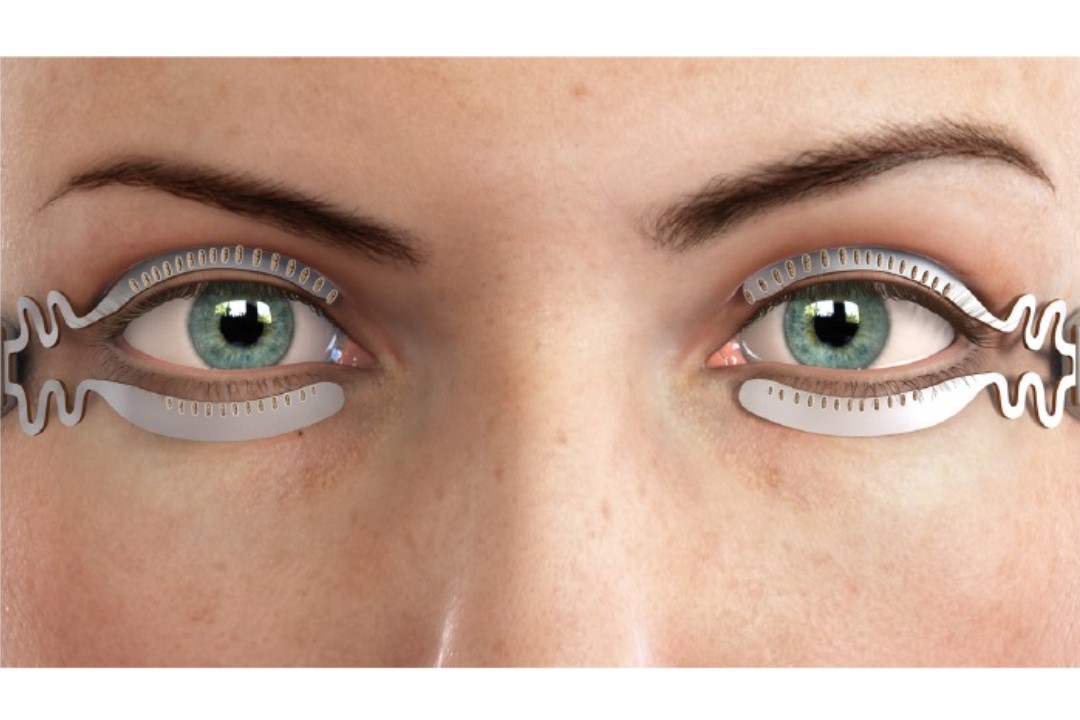
Sight Sciences’ TearCare system for the treatment of meibomian gland dysfunction (MGD) has received the US Food and Drug Administration’s (FDA) 510(K) device clearance.
Designed for adult patients with evaporative dry-eye disease due to MGD, Sight Sciences said the TearCare System delivers therapeutic heat to the outer surface of the eyelids to achieve the inner eyelid temperature of 41-42C necessary to melt meibum. The FDA’s pre-market approval follows safety and effectiveness data from the Olympia study, which also demonstrated the device, used in conjunction with manual gland expression, significantly improved patients’ mean tear film breakup time and meibomian gland secretion score.
For more, see https://eyeonoptics.co.nz/articles/archive/dry-eye-research-review/